The kids will get a lesson about osmosis with this science experiment using gummi worms!
Time: Two days
Skill Level: Medium
Supplies
4 gummi worms or other gummi candy
3 small clear bowls, each with 1 cup of water
Sugar
Salt
Directions
1. Add a spoonful of sugar to one bowl of water, and a spoonful of salt to another bowl of water.
2. Put a gummi candy in each bowl (set extra gummi candies aside for comparison).
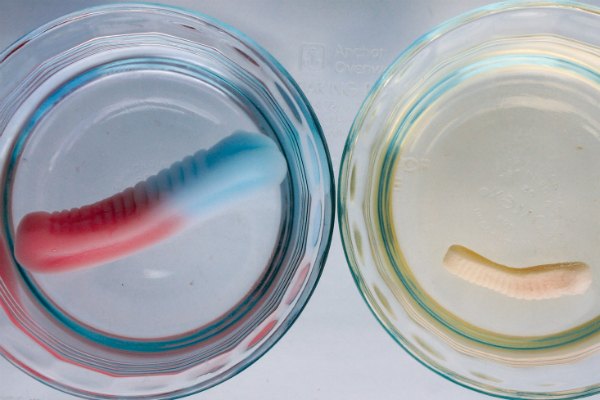
3. After several hours, check the gummi candies to see what size they are. Which ones have changed the most?
What’s happening:
A cup of fresh water is a dilute solution. When you put salt in water, you make a concentrated solution. The more salt, the more concentrated the solution is. If water can pass from a dilute solution into a concentrated one, it will. Nature is trying to make the two solutions equally concentrated. This process is called osmosis.
You can see osmosis in action when you put a gummi worm in fresh water. Water flows into the gummi worm, diluting the sugary gelatin mix (a concentrated solution). The gummi worm expands. But if you put a gummi worm in salt water, the salt water is already concentrated. Not as much water is needed to dilute the gummi worm, so it will expand less. Depending on how concentrated your solution is, it might not expand at all.
Osmosis is also the process that draws water into plant roots and up where it’s needed. If you watered plants with salt water, the plant cells wouldn’t be able to absorb any water, just like the gummi candies.
Science fair ideas
Mix up different solutions with different substances and put a gummi worm in each. Predict which gummi worm will expand the most and which will expand the least.
----------
Find more experiments and science fair ideas in the book, Candy Experiments 2 by Loralee Leavitt/Andrews McMeel Publishing, LLC.
Pubished: March 2015














